 
This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Henry Agnew, and Lance S. Tantalum is the 73rd element of the periodic table so its atomic number is 73. The atomic mass unit (amu) is a unit of mass equal to one-twelfth the mass of a carbon-12 atom Read More Protons, Neutrons, Electrons for Cesium (Cs, Cs+) Protons, Neutrons, Electrons for Tantalum (Ta, Ta5+) Tantalum is a classified transition metal and its symbol is ‘Ta’.Both protons and neutrons have a mass of 1, while electrons have almost no mass. The negative charge of one electron balances the positive charge of one proton. As a result, a neutral atom must have an equal number of protons and electrons. As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. It is a very ductile, pale metal, which darkens in the presence of trace amounts of oxygen. Of all elements that are solid at room temperature, caesium is the softest: it has a hardness of 0.2 Mohs. The positive charge on a proton is equal in magnitude to the negative charge on an electron. In this video we’ll use the Periodic table and a few simple rules to find the number of protons and electrons for the Calcium ion (Ca2+). Physical properties High-purity caesium-133 stored in argon.Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron).Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force. It is found at Bernic Lake in Manitoba, Canada. It is highly explosive when exposed to water. It is the least abundant of the five non-radioactive alkali metals. Neutrons are a type of subatomic particle with no charge (they are neutral). Cesium sulphide (Cs 2 S) Cesium telluride (Cs 2 Te ) Interesting facts.Protons are bound together in an atom's nucleus as a result of the strong nuclear force. 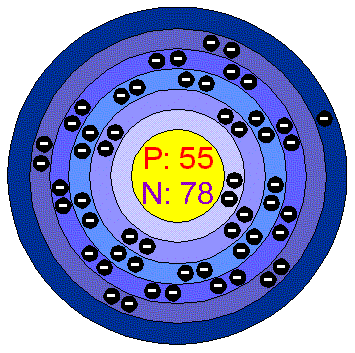
Protons are a type of subatomic particle with a positive charge.An atom of Caesium has only 1 electron in. Electrons are a type of subatomic particle with a negative charge. Caesium as 55 protons and 78 neutrons in its nucleus giving it an Atomic Number of 55 and an atomic mass of 133.\): Properties of Subatomic Particles Particle 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
